 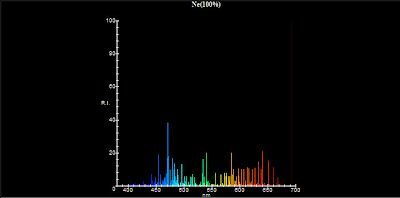
Atoms and molecules can also absorb and emit lower frequency, IR radiation. Research: Chaos, Semiclassical Mechanics, and Symmetry ViolationĬhristopher Keane Vice President for ResearchĮ-mail: Computational and experimental research in x-ray lasers, inertial confinement fusion, and ultra-high intensity laser-matter interaction. These atomic emission spectra can be used (often informally by using a flame test) to gain insight into the electronic structure and identity of an element. Emission spectroscopy examines the wavelengths of photons emitted by. See how the characteristic spectra of different elements are produced, and configure your own elements. Stock photo The atomic emission spectra of Neon gas. Other lines are located at 834.761 nm, 878.062 nm, and 885.387 nm. Produce light by bombarding atoms with electrons. Research: nuclear many-body theory to answer outstanding questions about superfluids in nuclei, neutron stars, cold atoms condensed matter and atomic physics high energy physics (QCD) with applications to astrophysics and cosmology computer science The most prominent line in the emission spectrum of neon is found at 865.438 nm. 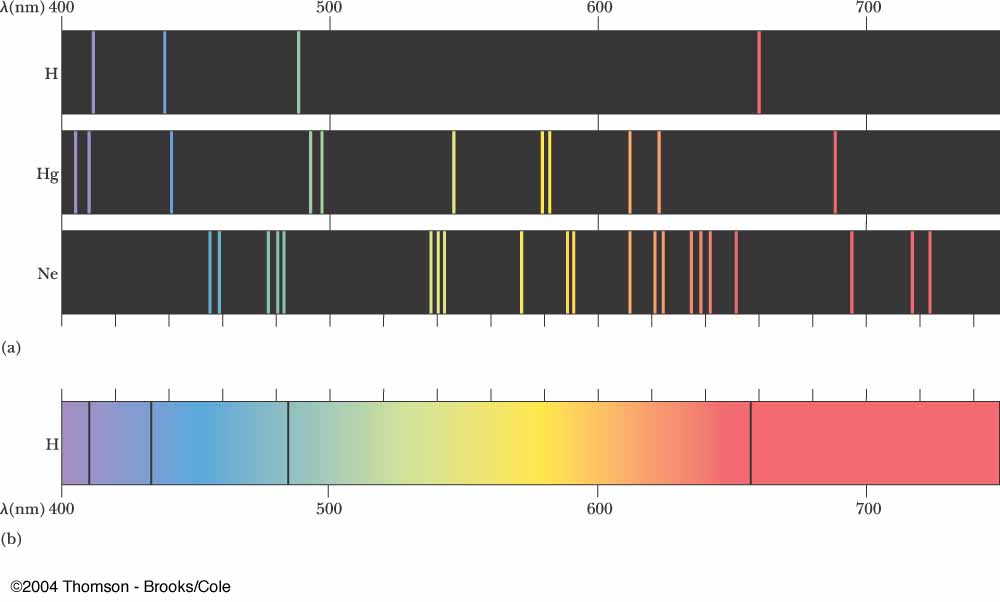
Michael McNeil Forbes Apm.forbes Michael McNeil Forbes Associate Professor of Physics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
